IQ, OQ, and PQ services for GMP and GDP
One-stop for your entire validation process
Get your validations done faster with Eupry’s qualification services built for GMP and GDP.
- One provider for the full validation
- Cost- and time-optimized process
- Full GxP compliance confidence
Get an instant overview of all the service options and technical specifications.
Trusted by 1000+ pharma and healthcare logistics companies worldwide including:
One provider, everything you need
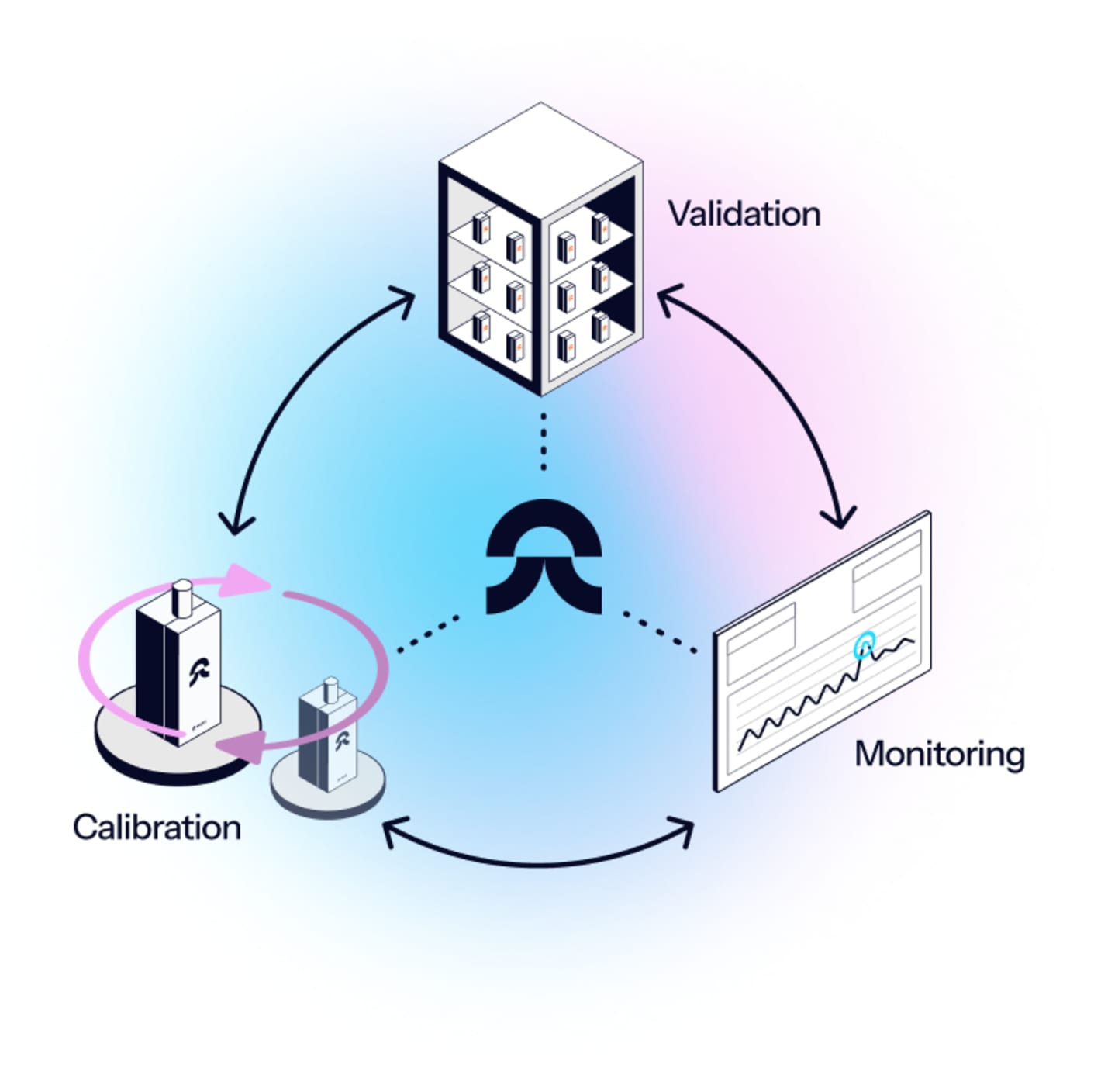
Eupry’s validation services cover the full qualification cycle: Installation qualification (IQ) to operational qualification (OQ) and performance qualification (PQ).
Let us handle the complete qualification … or pick and choose the parts that make sense for your operations.
See all the solution options instantly.
Why work with Eupry for IQ, OQ, and PQ?
- GxP compliance confidence: Thermal compliance is what we do. Get complete peace of mind that your units or facilities are fully qualified and operating correctly from the start.
- All you need for thermal compliance: From the initial IQ to the final PQ and continuous monitroing, our specialized solutions cover all you need.
- Minimized costs and time-spent: Cut costs with one vendor and one (unified, cost-optimized, and automated) solution that includes calibration and a full warranty.


Trusted by 1000+ companies worldwide

Download a free IQ/OQ/PQ solution catalog
Ensure reliable temperature validation for GDP and GMP - in less time. See how it works in the free catalog.
Specialized in (pharma) validation
Meet the standards of GMP and GDP and take your units into operation – in less time.
With thousands of validations under our belt, we know how to ensure your temperature-controlled equipment meets industry standards.
Our services and equipment are designed especially for GxP temperature compliance.
= GxP-reliability, faster

Short version: What is the solution about?
- What: One specialized service covering the entire thermal validation process from IQ to OQ and PQ.
- Why: To make the process as reliable and efficient as possible.
- For who: Highly regulated industries like pharma, biotech, and pharmaceutical logistics.
- For what: Any temperature-controlled unit or facility – from freezers and incubators to warehouses.

How the services work
Our validation team handles the full IQ/OQ/PQ process of your temperature-controlled system.
Depending on your requirements, the services can include:
- Installation qualification (IQ): Our team verifies that the unit is installed correctly according to the manufacturer’s specifications.
- Operational qualification (OQ): We ensure that the unit meets all URS requirements, such as maintaining the required conditions and triggering alarms, including empty temperature mapping studies.
- Performance qualification (PQ): This process validates the unit’s performance under actual working conditions, often through loaded mappings.
Tip! The mapping studies are tailored to fit your needs – from our full-service option, where our team handles everything, to a more hands-on approach using our mapping kit. Either way, you will have our team’s guidance every step of the way.

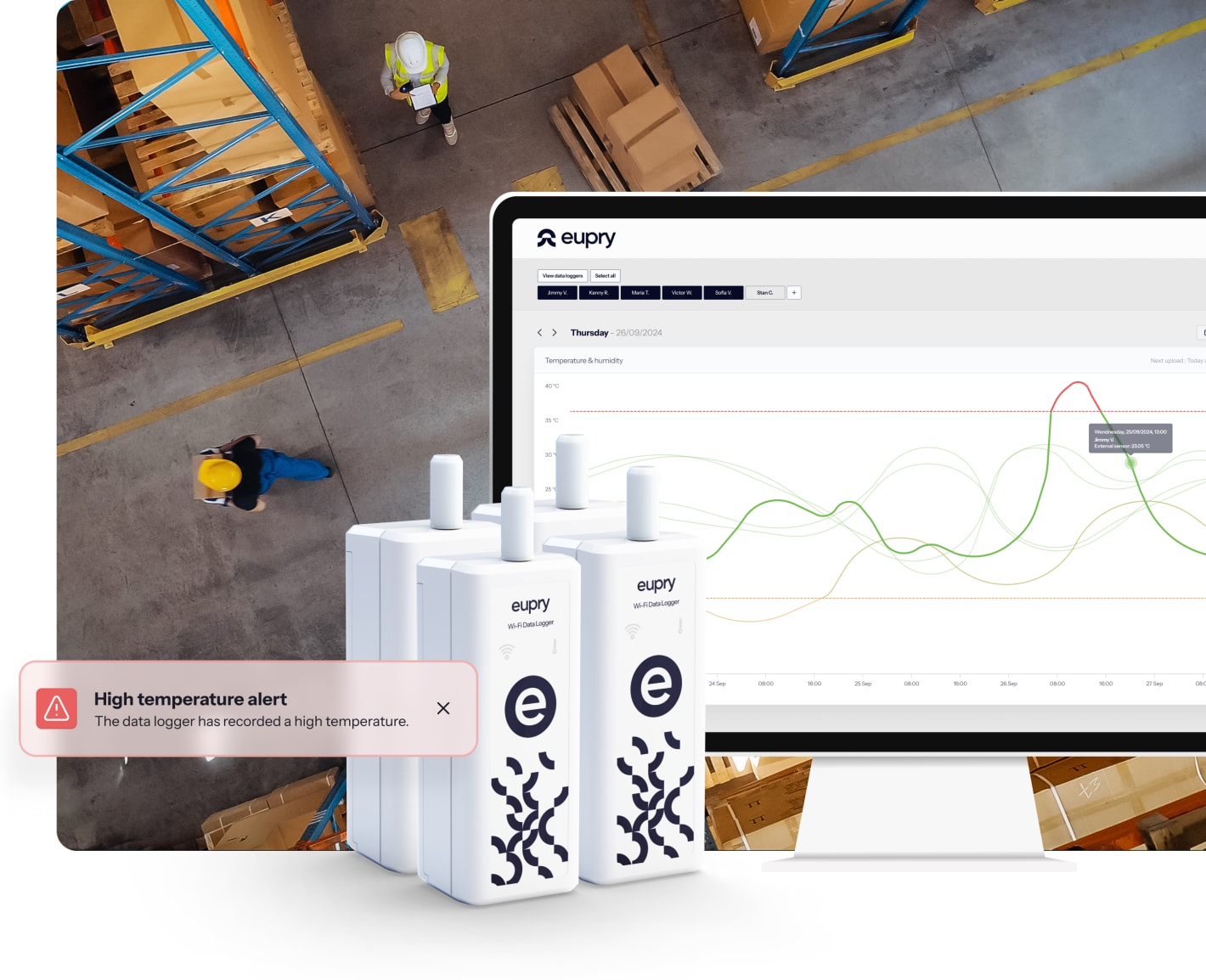
The fastest calibration solution you will find included
With Eupry's on-the-wall calibration solution, you can handle all calibrations on-site and in minutes, and thereby, reduce the time you spend on calibration by up to 95%.

Turn validation into your competitive edge
- Superior quality and GxP confidence
- The most cost-efficient solution
- Minimized operational downtime
With Eupry, temperature validation goes from operational roadblock to competitive advantage.
Dependable: We proactively update our processes to meet the latest FDA and EMA standards, so you stay compliant.
Efficient: One provider for the full validation means fewer delays and faster equipment readiness.
Continuous: With continuous mapping and monitoring (CMM), you eliminate re-mappings and their costs, work, and downtime.

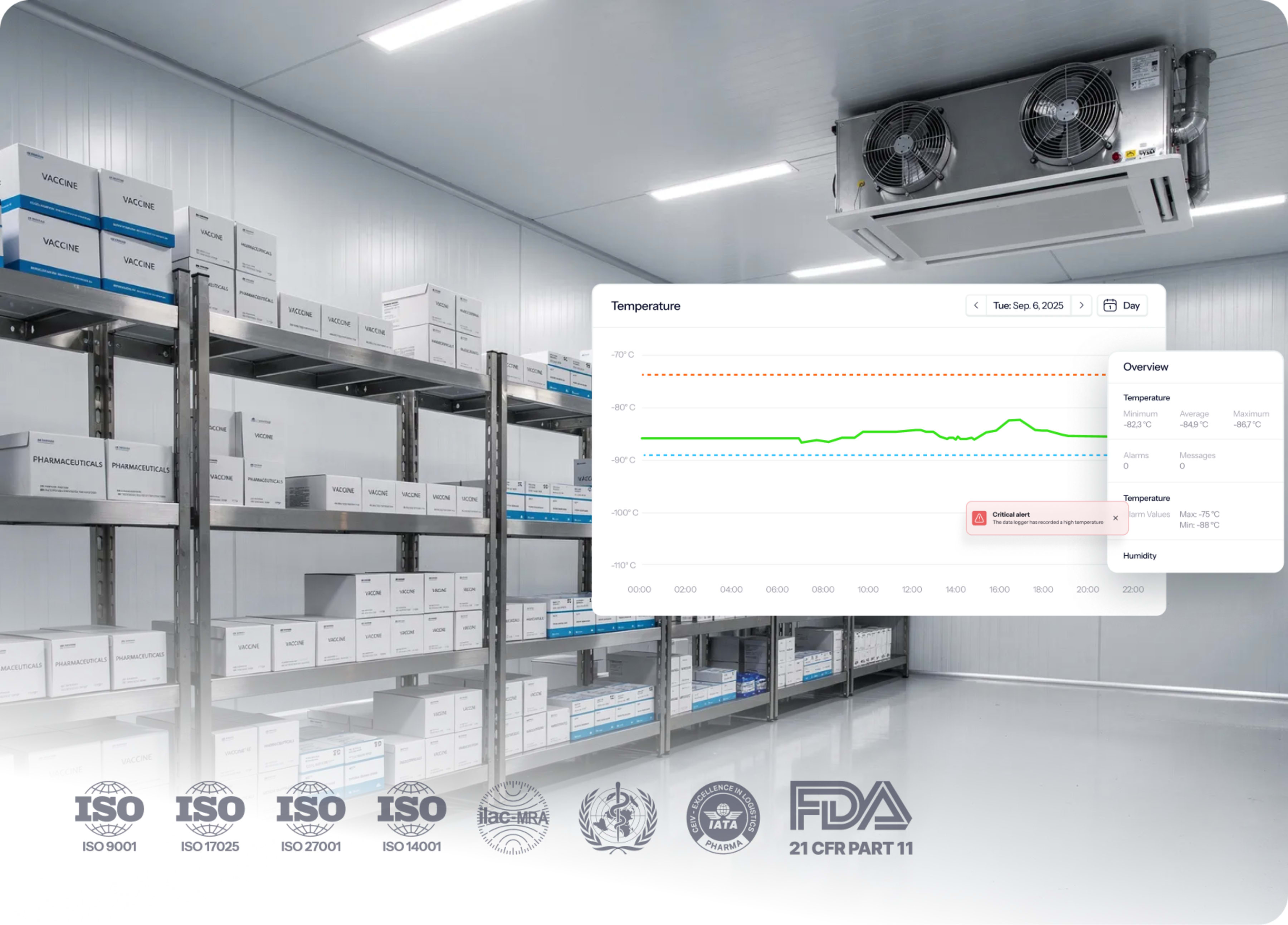
Everything you need for temperature compliance
What about after validation?
We have got you covered. Our automated thermal compliance solution allows you to easily transfer to ongoing monitoring with just a few clicks.
And (the market’s most efficient) calibration is included.
Once the validation is done, you simply:
- remove the extra data loggers.
- switch from mapping to monitoring software with a few clicks.
And you are done.