Continuous mapping (and monitoring) in practice
How pharma logistics companies are implementing CMM across global networks
Note: The talk is from Eupry's presence at the 22nd Temperature Controlled Logistics in Biopharmaceuticals Europe, 2026.
The speaker, Jakob Konradsen, is the co-founder and Chief Quality Officer at Eupry.
Temperature mapping has remained a point-in-time snapshot for decades...
– but that is changing.
Regulators increasingly expect continuous verification, and organizations managing multiple facilities face mounting pressure to demonstrate consistent quality control across their operations.
Continuous mapping and monitoring (CMM) turns validation into a constant state.
But how does it work?
In this session, Jakob Konradsen walks through:
- Examples of real CMM implementations in GDP
- What worked (and what failed)
- The concrete business value it brought
- Learnings and pitfalls to account for

Learn more about CMM

On-demand webinar: Continuous mapping
Watch the on-demand webinar to explore the framework, its use cases, pitfalls, and more.

Guide: Continuous temperature mapping in GxP
Get a step-by-step guide to implementing continuous temperature mapping in your operation.
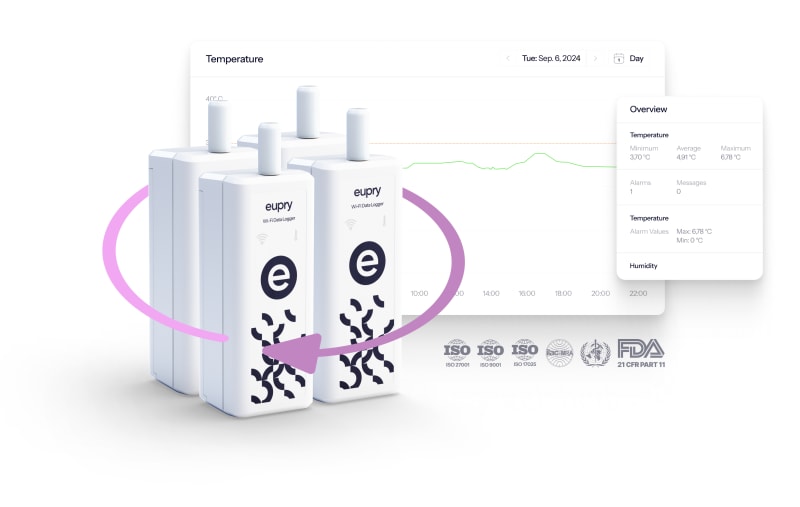
Solution: Continuous mapping (and monitoring)
Eupry’s CMM solution covers all your mapping and monitoring requirements. See how it works.